|
For example, a sample with a volume concentration of 25 µl/l will have a mass concentration of 25*2.65 = 66 mg/l. For mineral grains (clay, silt and sand sizes), this will typically be 2.65 g/cm3. To convert from ppm by volume to ppm by mass, multiply by the density of the particles. However, if ppm is expressed as THE MASS of particles in a unit volume of water, then ppm BY MASS is equal to mg/l. If ppm is expressed as THE VOLUME of particles to a unit volume of water, then ppm BY VOLUME is equal to µl/l.   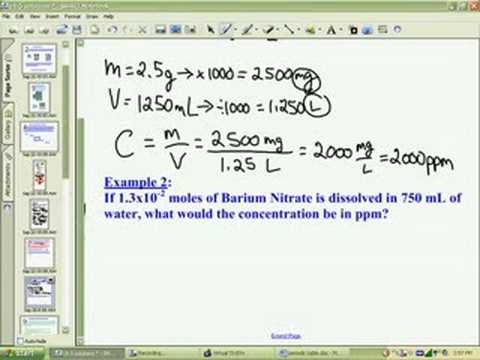
But what parts? The amount of particles in a suspension can be expressed as the total volume OR total mass of particles in a unit volume of water AND THESE TWO NUMBERS WILL ONLY BE THE SAME IF THE DENSITY OF THE PARTICLES IS 1 g/cm3.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
